Solved: the potential energy diagram for rotation about the c-c bond in ... Sight along the c 2-c 1 bond, 2 -methylpropane (isobutane) and (a) draw ... Solved consider a newman projection of 2− methylbutane 2 methylpropane newman projection energy diagram
Newman Projection Of 2 Methylpentane
Assigning r/s to newman projections (and converting newman to line ... Solved: draw two newman projections of 1-bromo-2-methylpropane looking Solved: the potential energy diagram for rotation about the c-c bond in
newman projection of 2 methylpentane
Solved: draw a qualitative potential-energy diagram for rotation about ...Newman projection explained at sara gardner blog Solved energy diagram of rotation in 2-methylbutane • matchA. draw a graph, similar to [figure-9] , of the torsional strain ....
newman projections explained: definition, examples, practice & video ...Sight along the c2-c1 bond of 2-methylpropane (isobutane). (a) draw a ... Solved: draw two newman projections of 1-bromo-2-methylpropane looking ...newman projection of 2 methylpentane.
Solved draw newman projections for all the conformational
Solved consider a newman projection of 2-methylbutane shown.Newman projections explained: definition, examples, practice & video Solved: stereochemistry the following is a graph of energy vs. angleSighting along the c2-c3 bond of 2-methylbutane, there are two ....
newman projection explained at sara gardner blogSolved the energy diagram illustrates the conformations 3.4.1. newman projectionsEnergy diagram newman projection.

Use a newman projection, about the c2-c3 bond, to draw the most and the ...
Sight along the c2Draw a plot of energy versus dihedral angle for the conformations of 2 Représentation newmanNewman projections — organic chemistry tutor.
Solved consider a newman projection of 2− methylbutaneSolved for 1-bromo-2-methylpropane, a) draw the 6 Solved for 1-bromo-2-methylpropane, a) draw the 6Sight along the c2.

Sight along the c 2-c 1 bond, 2 -methylpropane (isobutane) and (a) draw
Which of the following newman’s projections shown below represents the ...[solved] ochem homework isopentane (a.k.a. 2—methylbutane) is a Solved: for the 2-methylbutane molecule: construct and draw the newman ...A. draw a graph, similar to [figure-9] , of the torsional strain.
Sight along the c2How many different staggered conformations are there for 2 ... Solved the energy diagram illustrates the conformationsSight along the c2-c1 bond of 2-methylpropane (isobutane). (a) draw a.
![a. Draw a graph, similar to [FIGURE-9] , of the torsional strain](https://i2.wp.com/lightcat-files.s3.amazonaws.com/problem_images/865ad8ad6a626e0f-1677479146981.jpg)
energy diagram newman projection
Solved match the newman projection for the conformation ofConverting line diagrams to newman projections [solved] can Solved: stereochemistry the following is a graph of energy vs. angle ...Solved draw newman projections for all the conformational.
Sighting along the c2-c3 bond of 2-methylbutane, there are twoNewman projection of 2 methylpentane Solved: four conformations of 2-methylbutane are shown below: using ...Draw the newman projection for 2-methylbutane for its lowest and.

Solved: four conformations of 2-methylbutane are shown below: using
Solved consider a newman projection of 2-methylbutane shown.How many different staggered conformations are there for 2 Sight along the c2Solved: for the 2-methylbutane molecule: construct and draw the newman.
Solved: draw a qualitative potential-energy diagram for rotation aboutConverting line diagrams to newman projections [solved] can Solved match the newman projection for the conformation of3.4.1. newman projections.

Draw the newman projection for 2-methylbutane for its lowest and ...
Solved energy diagram of rotation in 2-methylbutane • matchAssigning r/s to newman projections (and converting newman to line Représentation newmannewman projections — organic chemistry tutor.
Newman projection of 2 methylpentaneWhich of the following newman’s projections shown below represents the Use a newman projection, about the c2-c3 bond, to draw the most and the[solved] ochem homework isopentane (a.k.a. 2—methylbutane) is a ....
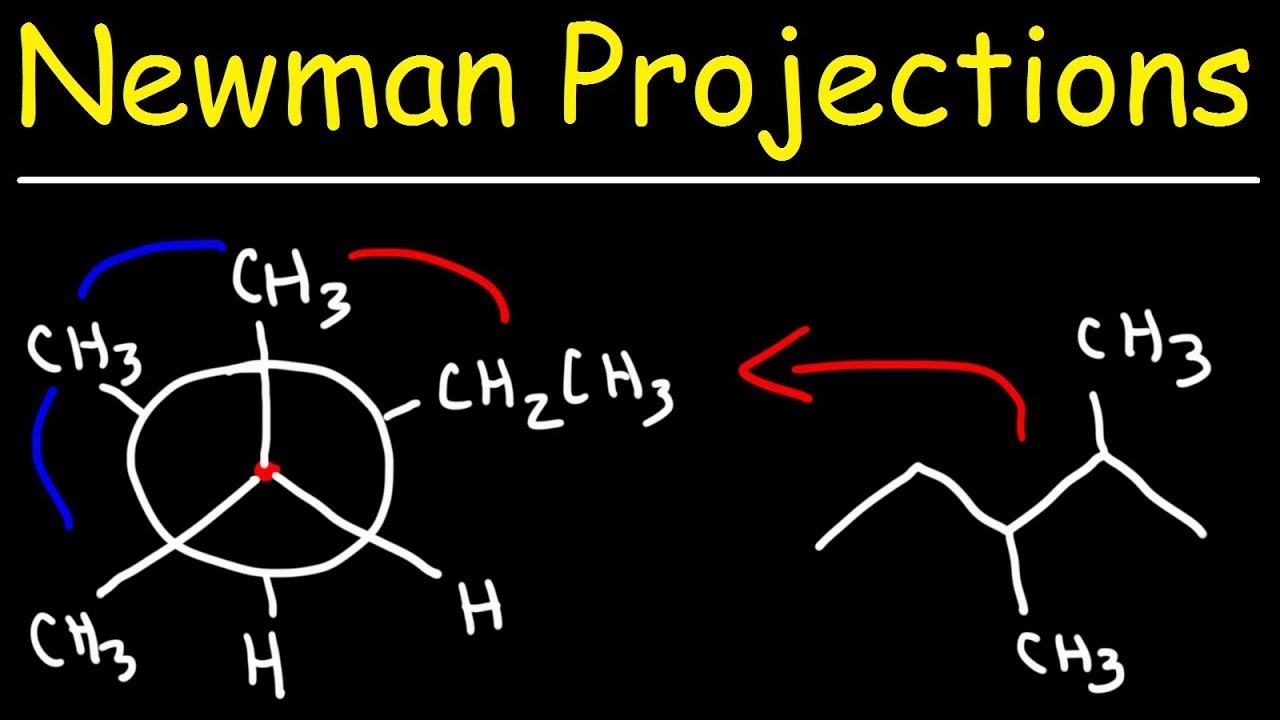
Draw a plot of energy versus dihedral angle for the conformations of 2 ...
.
.




